Materials—they are the basis upon which all things are made. New developments at every stage of their life cycle are the common thread to solving our energy, sustainability and environmental challenges.
Here are five of our most recent professors who are pushing boundaries in materials innovation to address some of today’s most pressing issues.
 |
||
| Purified solar-grade silicon | Mansoor Barati, Associate Professor & Gerald R. Heffernan Chair in Materials Processing |
Enabling grid parity for solar-based electricity
Silicon—it’s the second most abundant element in our Earth’s crust. Many of our products today are fabricated from silicon-enabled materials, including high-strength alloys in our cars and airplanes to semiconductors that make up our electronics and solar panels.
One major roadblock in taking photovoltaic power mainstream is its materials production cost. Professor Mansoor Barati and his group work on energy-efficient ways to make solar-grade silicon, lowering production costs to enable grid parity for electricity from our sun.
“There’s no doubt that our energy mix is moving towards renewable sources, such as solar power. Silicon is the backbone for large scale harvesting of the sun’s energy; the ability to effectively produce this material will allow us to generate electricity for mainstream use with the lowest environmental impact possible.”
 |
 |
|
| SEM image of nanoporous silica (SiO2) glass fabricated around a self-assembled polymer template |
Benjamin D. Hatton, Assistant Professor |
New materials with properties inspired by nature
Nature provides wonderful blueprints. Natural materials such as enamel, bone, mollusk shell, and plant leaves are examples of complex nano-composites that often have multiple structural and biochemical functions, and the ability to adapt to their environment.
Taking cues from these naturally-occurring materials and processes is what inspires Professor Benjamin Hatton’s research. He and his group investigate materials structures formed by self-assembly—a fabrication approach beyond the capability of conventional methods.
“I’m excited about the future of programmed self-assembly. The ability to control and organize complex 3D structures at the micro- and nano-scale can allow us to design materials with novel properties for applications such as switchable adhesion, triggered drug release, and self-cleaning anti-microbial surfaces.”
 |
 |
|
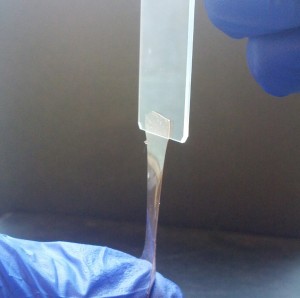
| Lian’s polymer electrolyte film |
Keryn Lian, Associate Professor |
Powering the future with polymers
Conventional batteries and super-capacitors use liquid electrolytes to conduct current, which require bulky and rigid structures for containment, and pose both safety and environmental hazards if it leaks.
Professor Keryn Lian has an answer to this problem—don’t use liquid. Instead, she and her group work on ionic conducting polymer membranes—an electrolyte film that conducts electricity in the solid-state, alleviating the need for containment structures and opening the door for a range of compact form factors.
“Polymer electrolyte films will allow us to develop technologies that are much safer and portable than ever before. Their lightweight and flexible attributes make them efficient in applications ranging from wearable electronics to automotive without adding extra volume from packaging.”
 |
 |
|
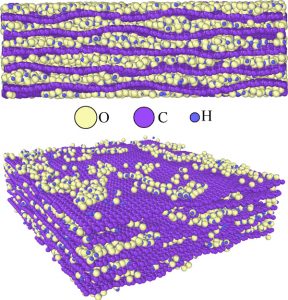
| Graphene oxide model comprised of oxygen and hydrogen groups bonded between carbon sheets |
Chandra Veer Singh, Assistant Professor |
Modelling 2D materials to chart new capabilities
Graphene — the world’s strongest material is also one of the lightest. As a one-atom thick layer, it is 100 times stronger and three times lighter than steel and can conduct both heat and electricity better than copper. Since its first synthesis over a decade ago, graphene research has expanded dramatically to tap into this wonder material’s potential.
Modelling graphene and other two-dimensional materials with a computational approach is the focus of Professor Chandra Veer Singh and his group’s efforts.
“The availability of high-powered computing has allowed us to investigate novel 2D atomic structures like graphene through simulation. This data can then aid in more controlled experimental approaches, accelerating the lab to commercialization process. We’re going to see new materials become available to us at a much faster pace.”
 |
 |
|
| SEM image of the zebra mussel adhesive apparatus |
Eli Sone, Associate Professor |
Bio-inspired adhesives and coatings
Zebra mussels are an invasive species that cause major problems for Great Lakes industries. They secrete protein-based adhesive plaques and threads that allow them to anchor on a range of surfaces. The buildup of these mussels on water-intake pipes, for instance, creates significant bio-fouling complications for freshwater resources in the region.
Professor Eli Sone’s group, in collaboration with Professor Hatton’s team, work to uncover the proteins used in this process in order to design anti-fouling coatings for adhesion prevention. They also investigate the mussels’ gripping mechanism in order to enable the design of bio-inspired adhesives for medical and dental applications.